
Loftware Cloud is the world’s first public, validation-ready Cloud labeling solution. It is the simplest way to design and print labels in a regulated environment.

Click the flashing orange dot below to move through the steps to see how Loftware can work for you.

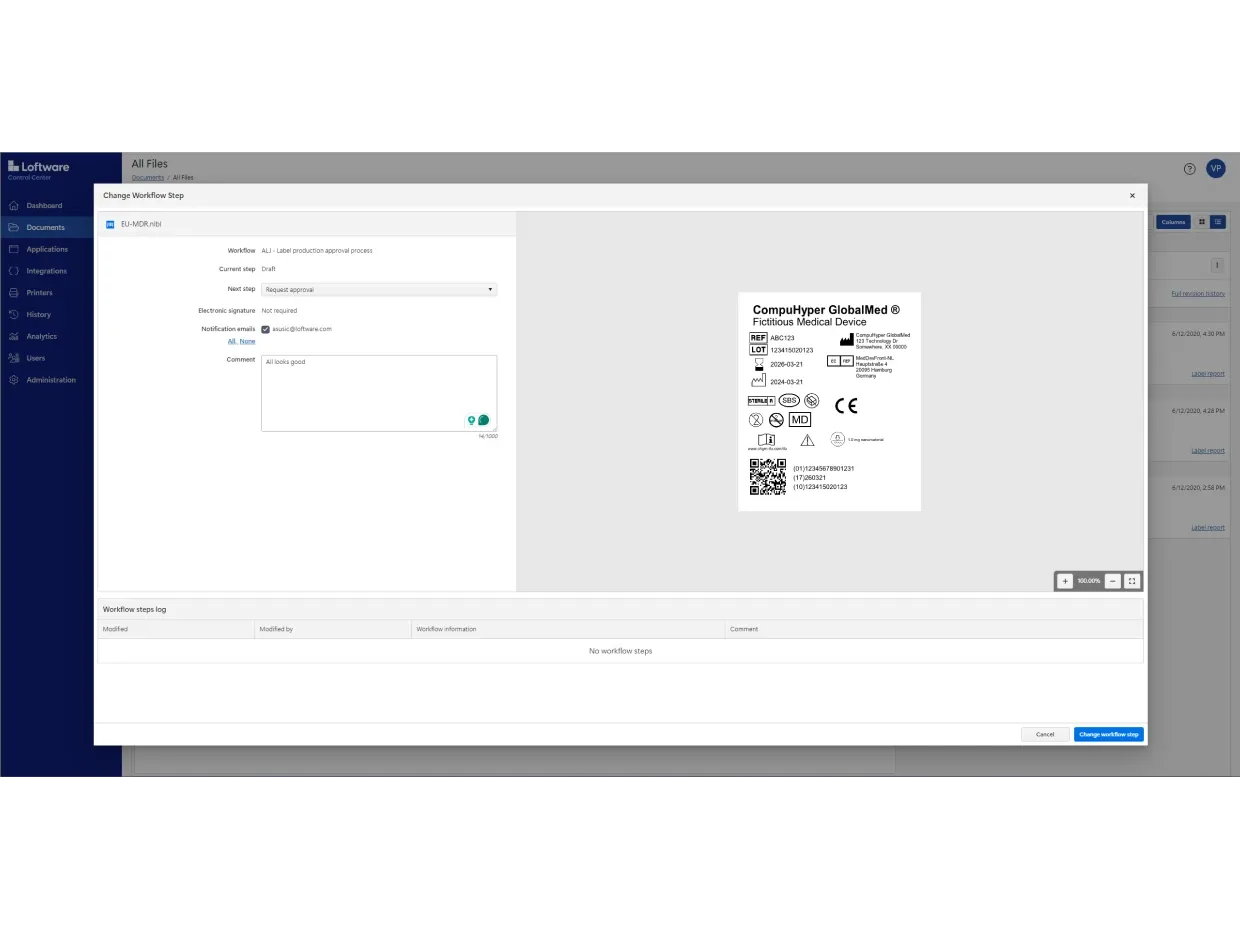
Use Loftware Cloud to digitize your entire approval process. You can also automate mass label changes and approvals without creating hundreds of label variations.
また、本番環境で稼動させる前に、システムの3層環境、すなわち開発環境(DEV)、品質保証環境(QA)、本番環境(PROD)を利用して、あらゆる変更をテストすることができます。
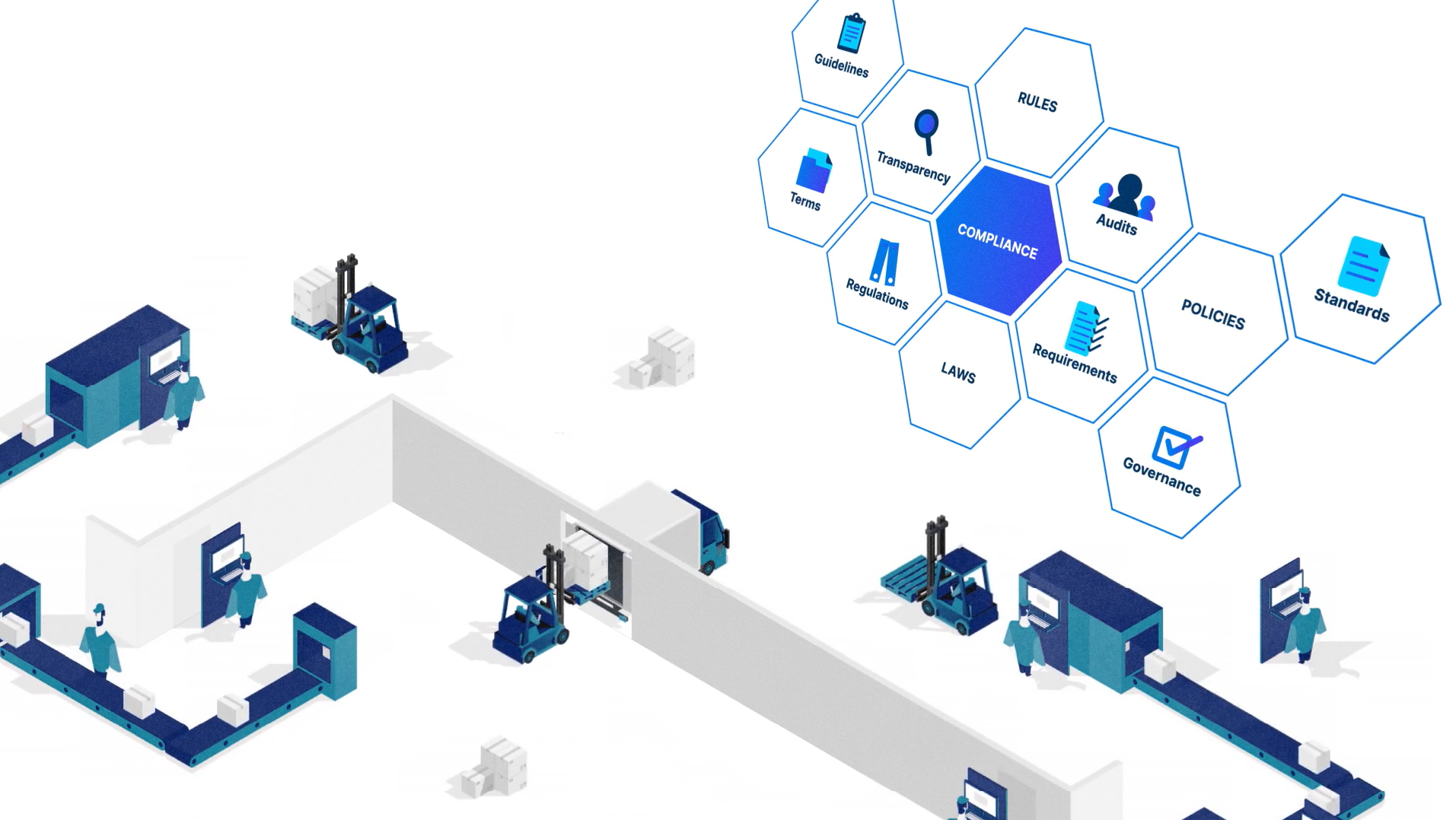
Loftware Cloud is designed to comply with the major regulations impacting the life science industry, including EU MDR, FDA UDI, FDA 21 CFR Part 11 and EU GMP Annex 11.
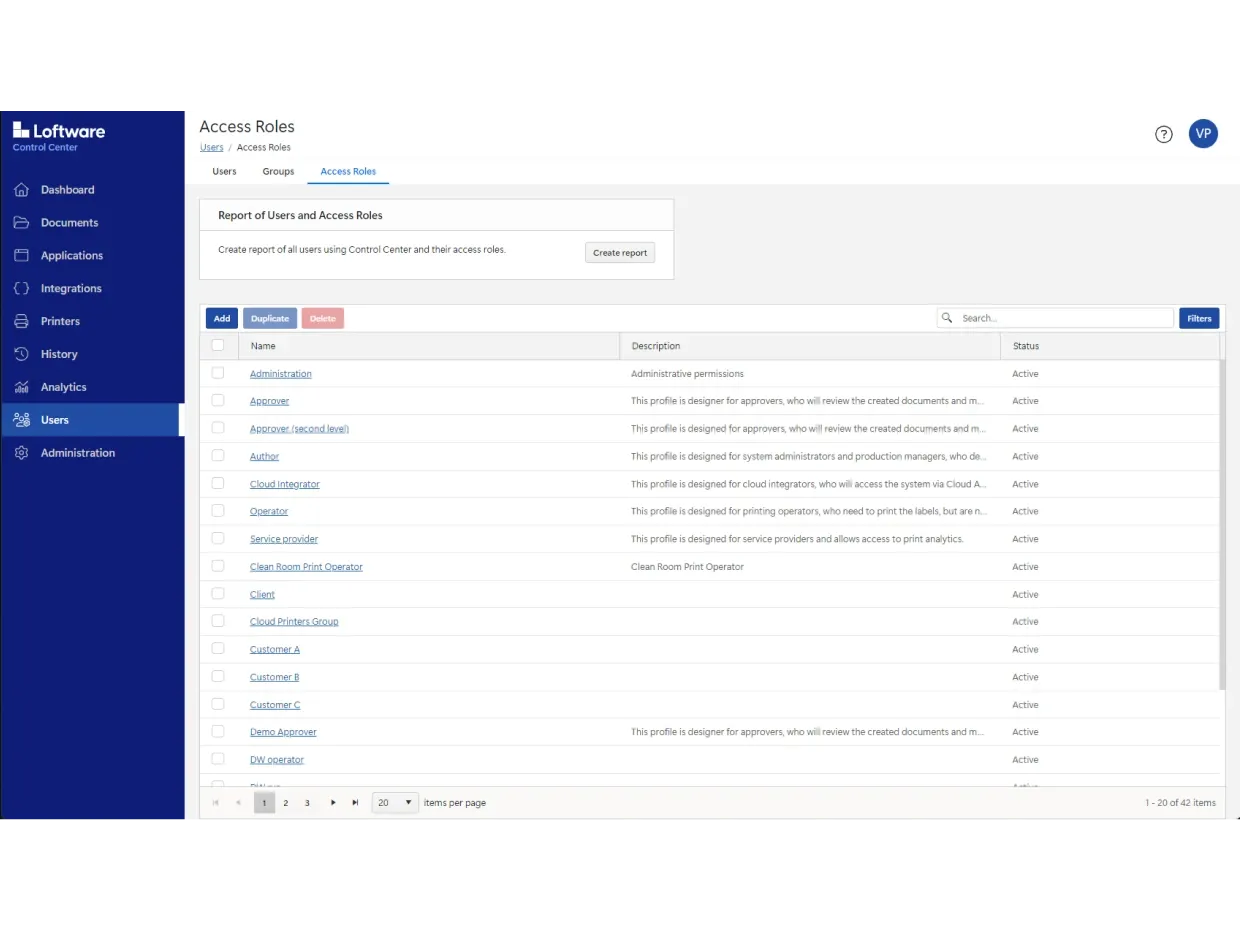
ロールベースのアクセス、文書のバージョン管理、設定可能な承認ワークフロー、電子記録・電子署名(ERES)が搭載されています。
また、12年間の印刷履歴も取得できるため、その間に印刷したすべてのラベルを視覚的に追跡できます。

Loftware Cloud is a validation-ready labeling solution. It is designed to streamline the validation process, which is further enhanced with the option of the Validation Acceleration Pack (VAP).
Our team can assist with IQ, OQ and PQ documentation as well. We also reduce the validation burden by only updating the software once a year. And when it’s time for the software release, we give you a three-month window before updating your production environment.
